Validating SaaS & Cloud Systems | Risk-Based GxP Compliance Guide

Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Validating SaaS and Cloud-Based Systems A Risk-Based Validation Guide for Regulated GxP Environments Introduction: The Shift to the Cloud Life Sciences organizations are rapidly transitioning from on-premise infrastructure […]
ALCOA+ Data Integrity Framework in CSV (2025) | FDA 21 CFR Part 11 Guide

Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X The ALCOA+ Framework: The Gold Standard for Data Integrity in Computer System Validation A complete 2025 guide to ensuring data integrity in CSV using ALCOA+ principles, FDA 21 […]
Transitioning from CSV to CSA in 2025 | FDA Computer Software Assurance Guide

Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Transitioning from CSV to CSA in 2025 | FDA Computer Software Assurance Guide Transitioning from CSV to CSA: A Practical FDA-Aligned Guide for 2025 Move beyond documentation-heavy validation. […]
The Definitive Guide to Computer System Validation (CSV) in 2025
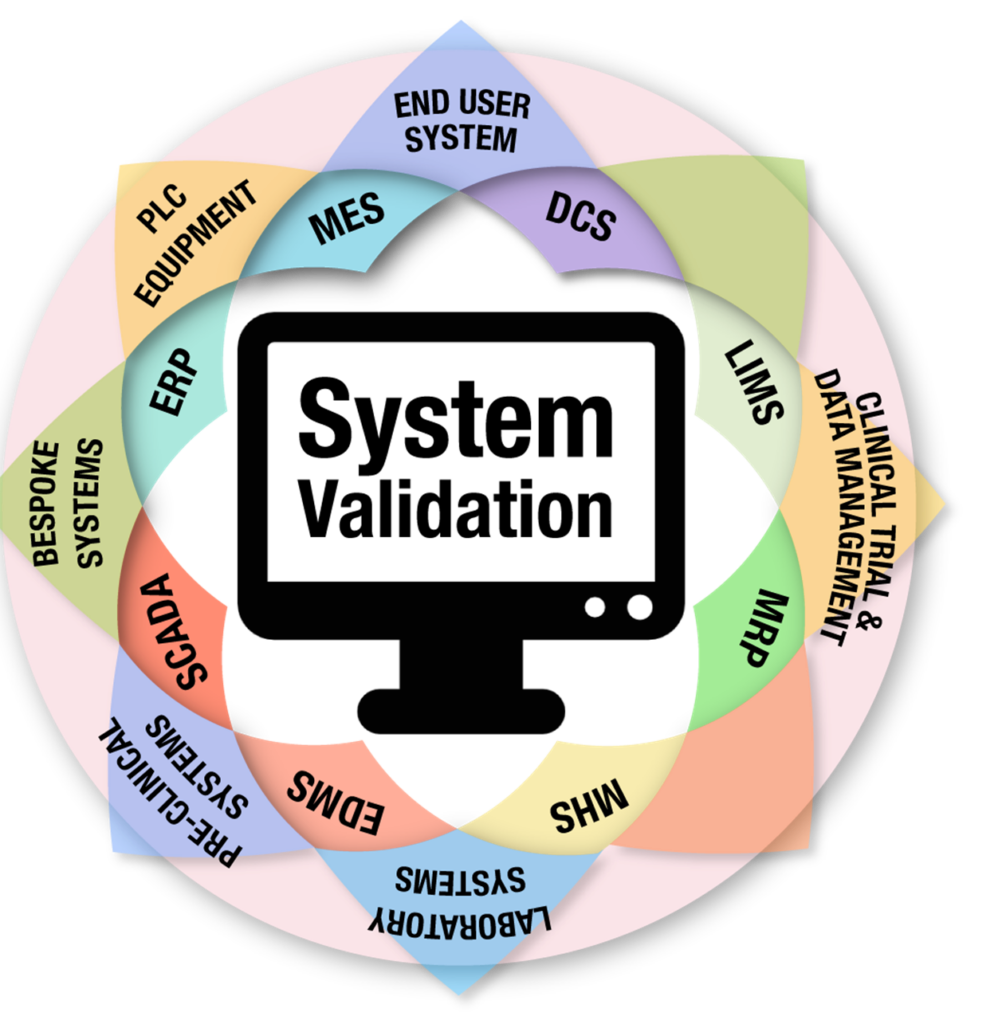
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Ultimate Guide to Computer System Validation (CSV) in 2025 | GAMP 5 & FDA Compliance The Ultimate Guide to Computer System Validation (CSV) in 2025 A complete compliance […]




