our blogs
Benefits of Electronic Quality Management Systems
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Why eQMS is the Future: Core Benefits of Electronic Quality Management Systems Explore why eQMS represents the future of quality management with core…
The Hidden Liability in Your Filing Cabinet
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X The Hidden Liability in Your Filing Cabinet Discover the hidden risks of paper-based Quality Management Systems (QMS) under FDA 21 CFR Part 11…
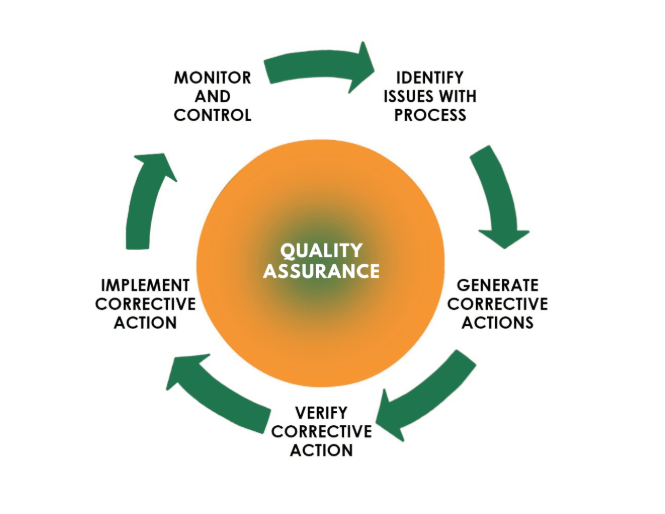
Mastering CAPA: The Engine of Continuous Improvement in Regulated Industries
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Mastering CAPA: The Engine of Continuous Improvement in Regulated Industries An exhaustive guide to Corrective and Preventive Action (CAPA). Learn the 7-step CAPA…
Integrating Risk-Based Thinking into the QMS
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Mastering CAPA: Continuous Improvement in Regulated Industries Published: December 26, 2025 Introduction: Why CAPA is the North Star of Your QMS A strong…
Quality Risk Management (QRM) and Supplier Quality Management (SQM)
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Mastering CAPA: Driving Continuous Improvement in Regulated Industries Published: December 26, 2025 Introduction: Why CAPA is the Heart of Your QMS A robust…
The Ultimate Guide to Equipment Life Cycle Management in Regulated Industries
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X The Ultimate Guide to Equipment Life Cycle Management in Regulated Industries Published: December 24, 2025 Introduction: Beyond the Asset Tag Equipment management is…
Predictive vs. Preventive Maintenance: Choosing the Best Strategy for Your EMS
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Predictive vs. Preventive Maintenance: Choosing the Best Strategy for Your EMS Introduction: Moving from “Fixing” to “Predicting” Equipment failures are among the most…
Calibration Management and Software Validation: Ensuring Accuracy and Compliance in Lab EMS
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Calibration Management and Software Validation: Ensuring Accuracy and Compliance in Lab EMS Introduction: The Science of Certainty in Digital Systems In regulated laboratory…
Audit Support & Inspection Readiness: A Strategic Blueprint for GxP Compliance
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Audit Support & Inspection Readiness: A Strategic Blueprint for GxP Compliance Published: December 24, 2025 Introduction: The High Stakes of Regulatory Inspections An…
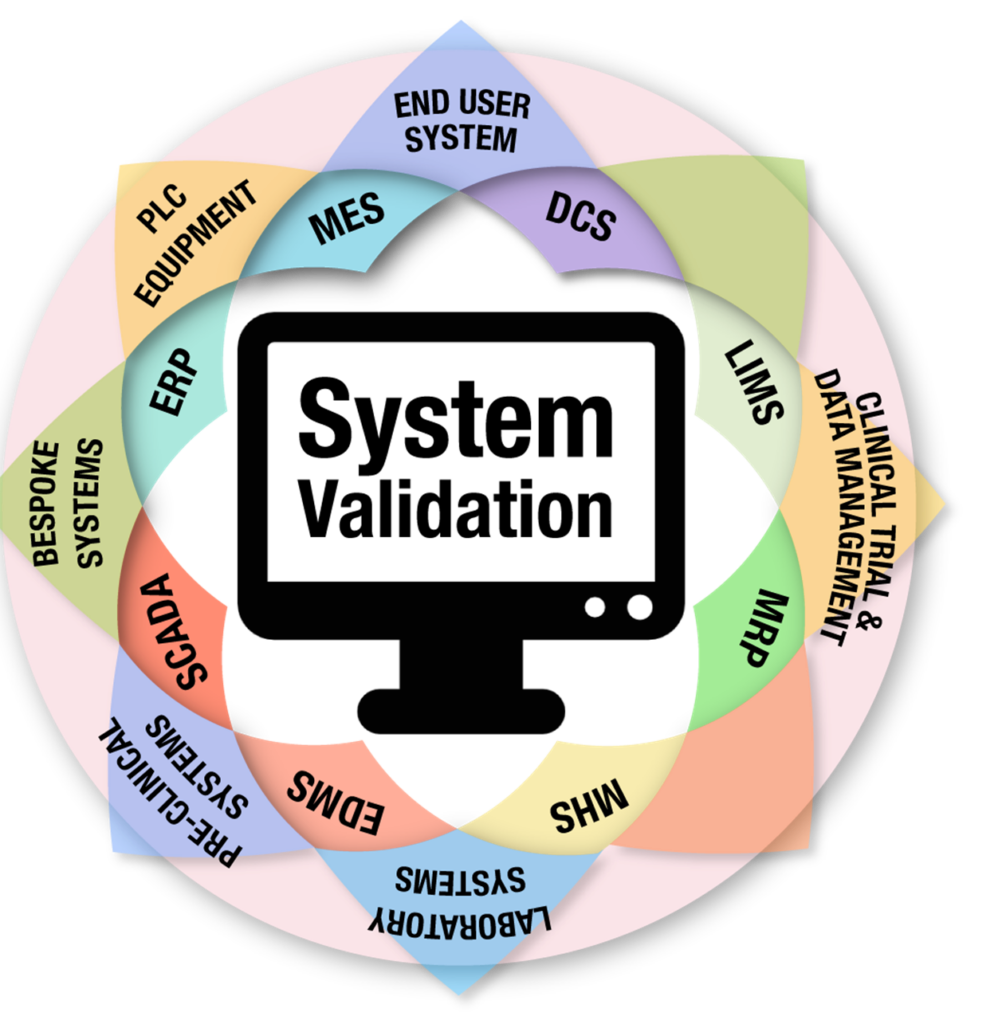
21 CFR Part 11 and Computer System Validation
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X 21 CFR Part 11 and Computer System Validation (CSV): Navigating the Digital Frontier Published: December 24, 2025 Introduction: The Virtual Quality Lab As…
Project Management and Quality Assurance in Life Sciences: Balancing Speed, Compliance, and Business Excellence
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Introduction: The Interplay of Speed, Compliance, and Quality Managing projects in Life Sciences requires more than traditional PM discipline. Success is measured not…
The Definitive Guide to Computer System Validation (CSV) in 2025
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Ultimate Guide to Computer System Validation (CSV) in 2025 | GAMP 5 & FDA Compliance The Ultimate Guide to Computer System Validation (CSV)…
Transitioning from CSV to CSA in 2025 | FDA Computer Software Assurance Guide
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Transitioning from CSV to CSA in 2025 | FDA Computer Software Assurance Guide Transitioning from CSV to CSA: A Practical FDA-Aligned Guide for…
ALCOA+ Data Integrity Framework in CSV (2025) | FDA 21 CFR Part 11 Guide
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X The ALCOA+ Framework: The Gold Standard for Data Integrity in Computer System Validation A complete 2025 guide to ensuring data integrity in CSV…
Validating SaaS & Cloud Systems | Risk-Based GxP Compliance Guide
Home About Us Services Learning and Development Contact-Us Our Resources Our Blogs Computerized System Validation (CSV) Services Quality Management System (QMS) Solutions Pharmaceutical Compliance Consulting Services X Validating SaaS and Cloud-Based Systems A Risk-Based Validation Guide for Regulated GxP Environments Introduction: The Shift to the Cloud Life Sciences organizations are…
Don't Miss Our Updates
Stay ahead with the latest updates, exclusive content, and insider news delivered straight to your inbox.

Bring us the problem — we’ll design the team and deliver the answer.
- 21 Castle Village Crescent, Celbridge, Co Kildare W23EW42, Ireland
- Support: +353873976644
- info@pegasuspharmaintl.com